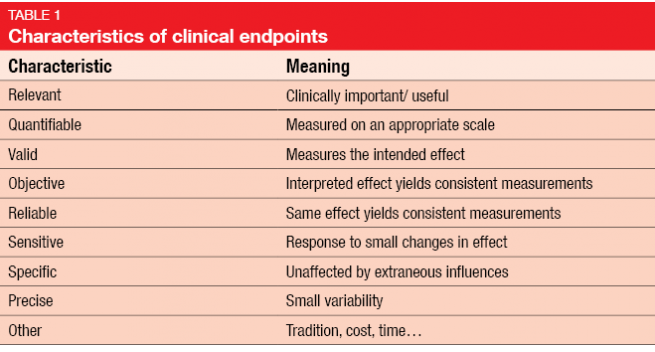
Development of Novel, Value-Based, Digital Endpoints for Clinical Trials: A Structured Approach Toward Fit-for-Purpose Validation | Pharmacological Reviews
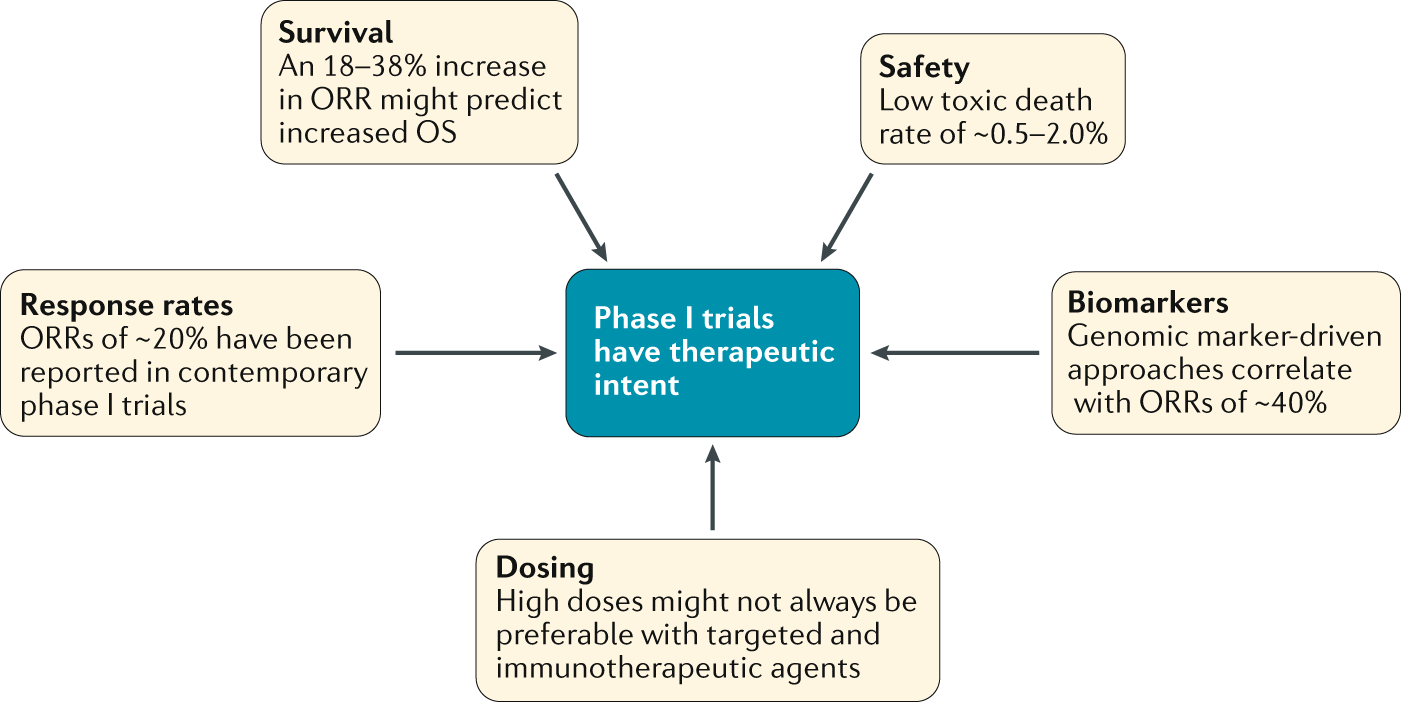
Phase I trials as valid therapeutic options for patients with cancer | Nature Reviews Clinical Oncology

A common language in neoadjuvant breast cancer clinical trials: proposals for standard definitions and endpoints - The Lancet Oncology

The Assessment of Clinical Trial Endpoints Along the Axes of Real-World... | Download Scientific Diagram
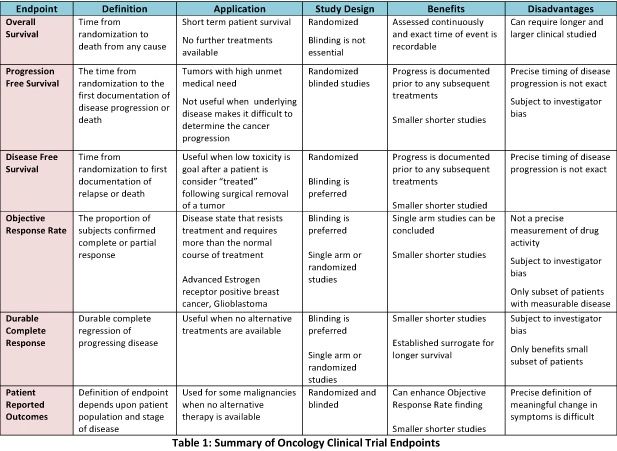
![PDF] The role of the U.S. Food and Drug Administration review process: clinical trial endpoints in oncology. | Semantic Scholar PDF] The role of the U.S. Food and Drug Administration review process: clinical trial endpoints in oncology. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f5e848a873b43037891286fb7c253ac50f83cc68/3-Table1-1.png)
PDF] The role of the U.S. Food and Drug Administration review process: clinical trial endpoints in oncology. | Semantic Scholar
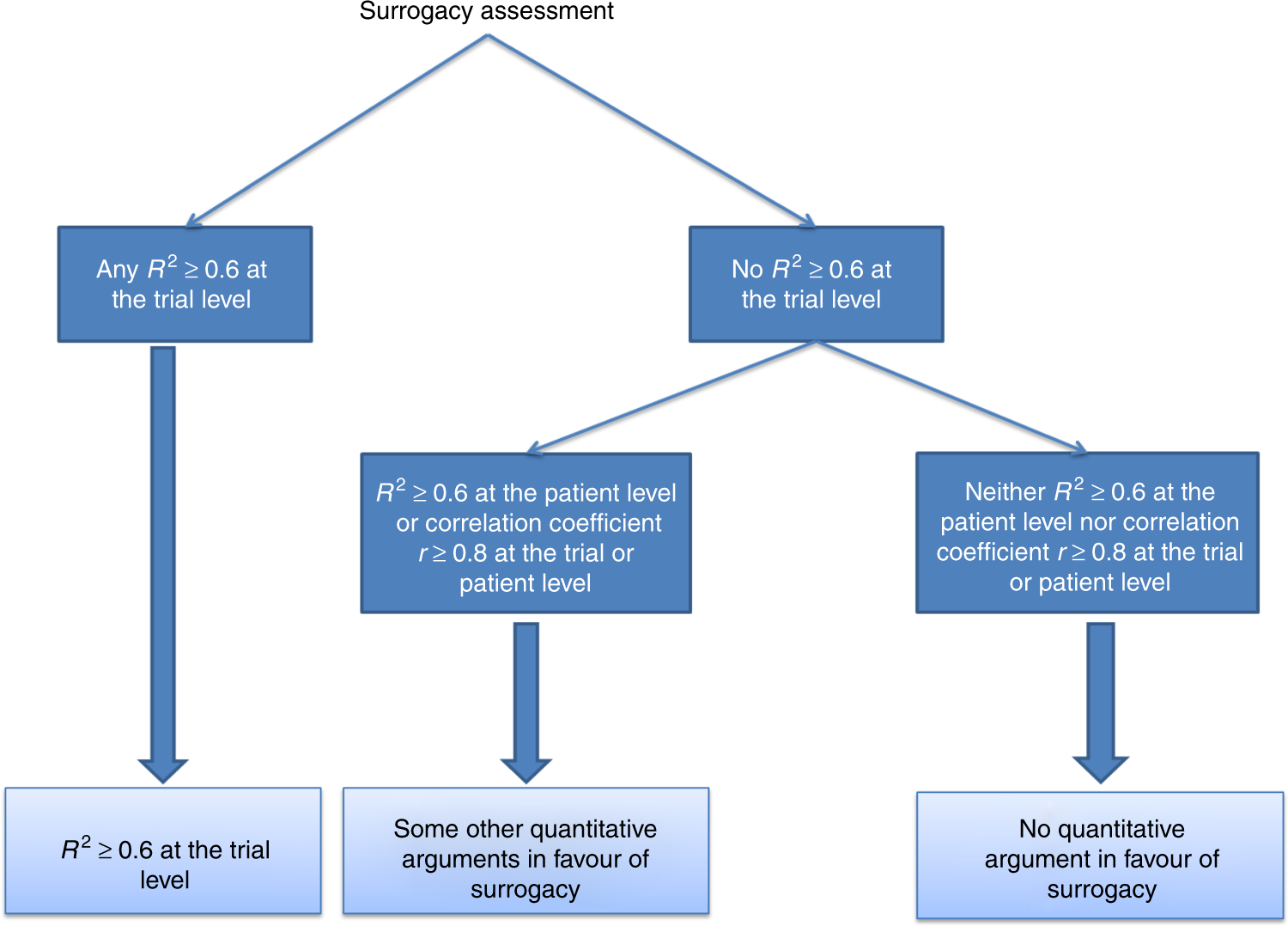
Progression-free survival as a surrogate for overall survival in oncology trials: a methodological systematic review | British Journal of Cancer

Functional and Symptomatic Clinical Trial Endpoints: The HFC-ARC Scientific Expert Panel | JACC: Heart Failure