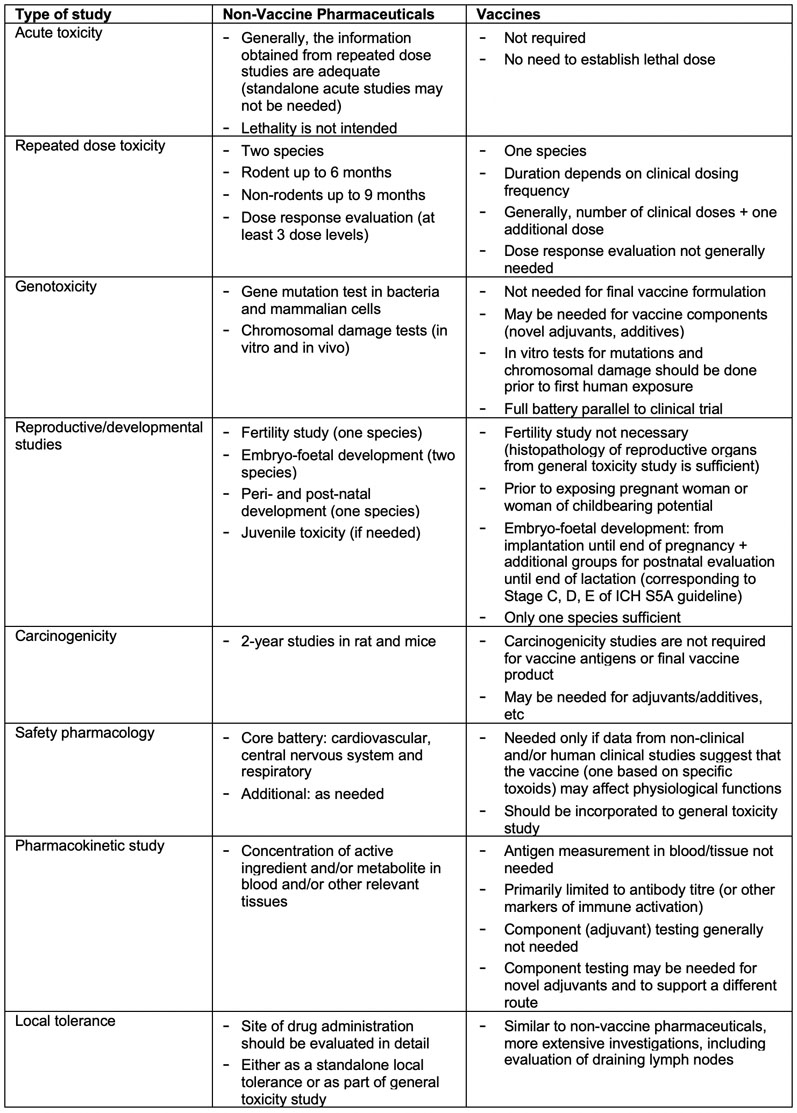
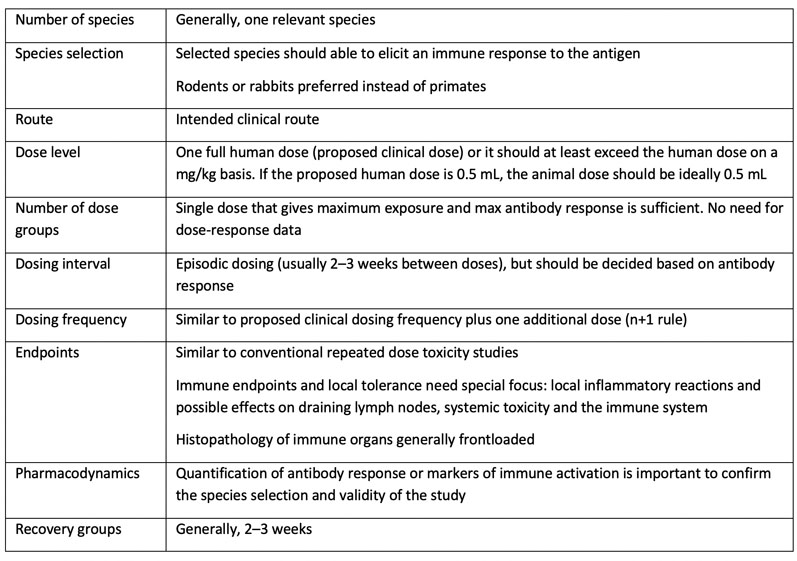
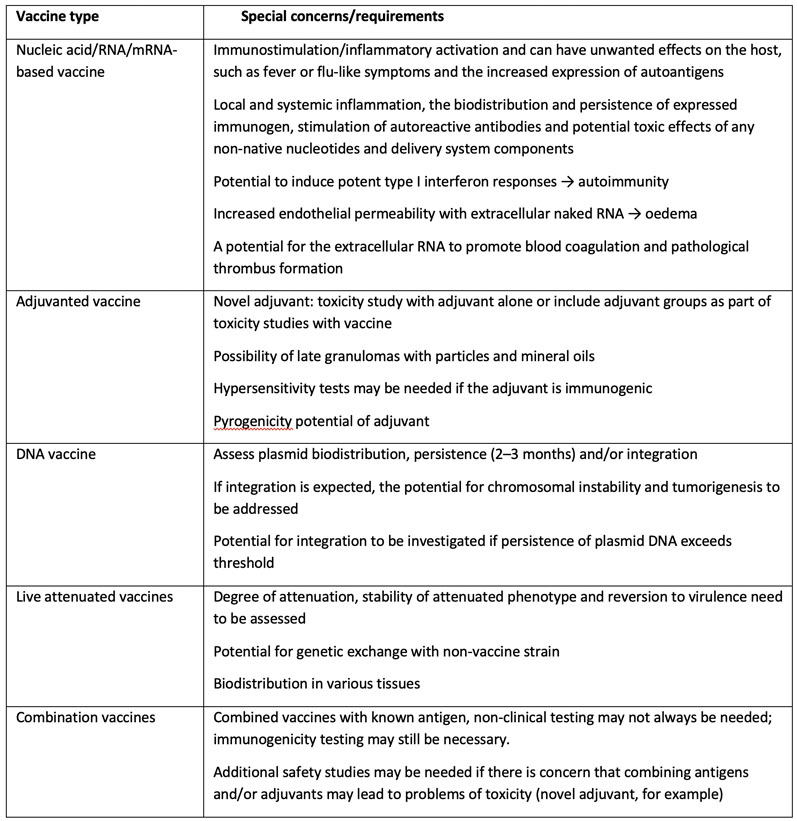
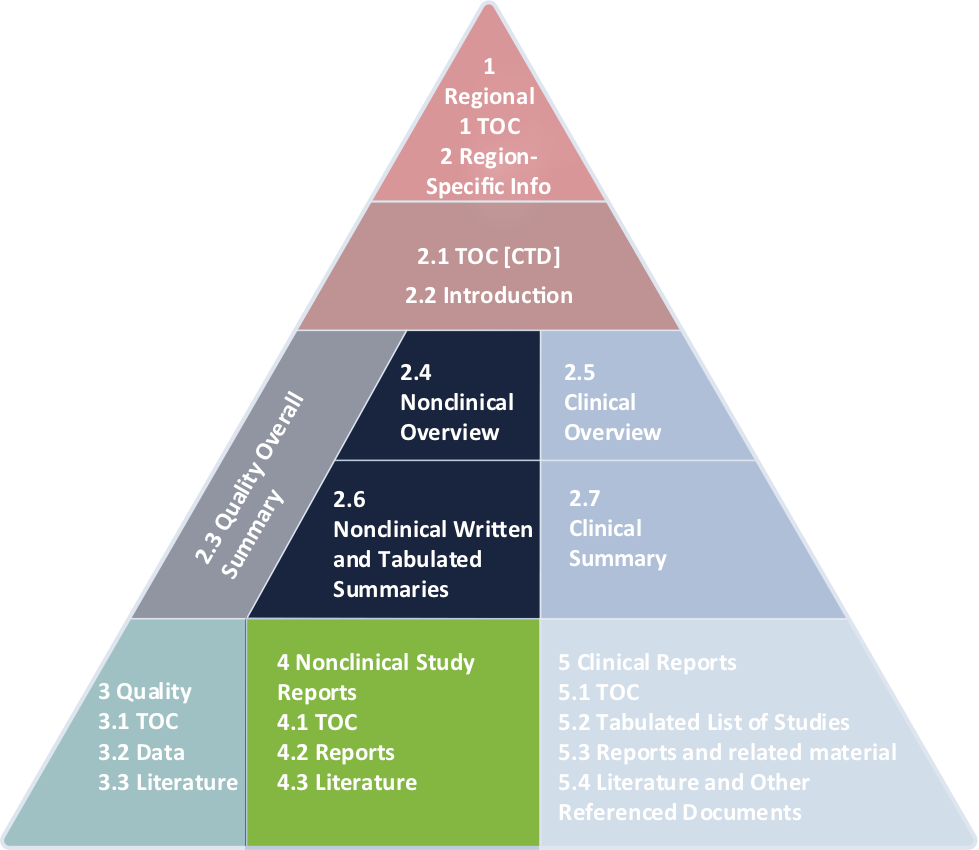
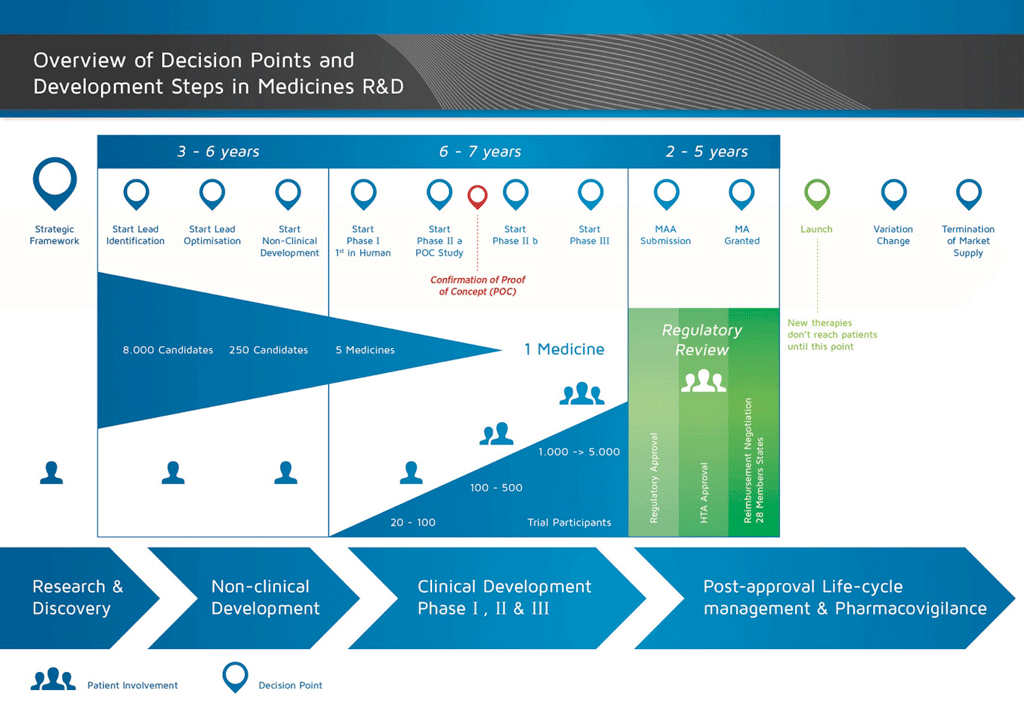
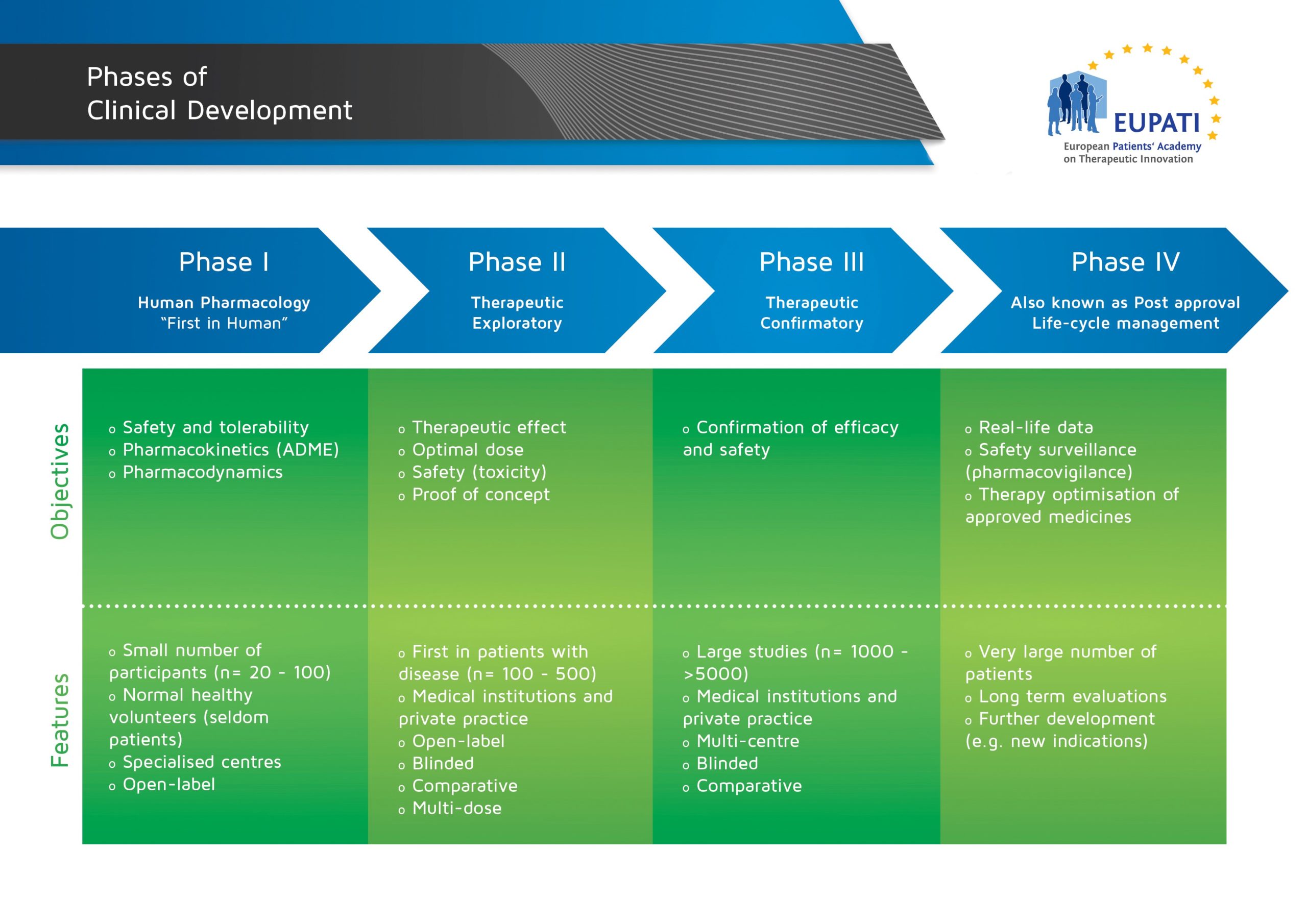
M3(R2) Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals

Non-clinical assessment of cell therapy products: the perspective from five Asian countries/regions based on regulatory guidelines and the underpinning rationales - Cytotherapy
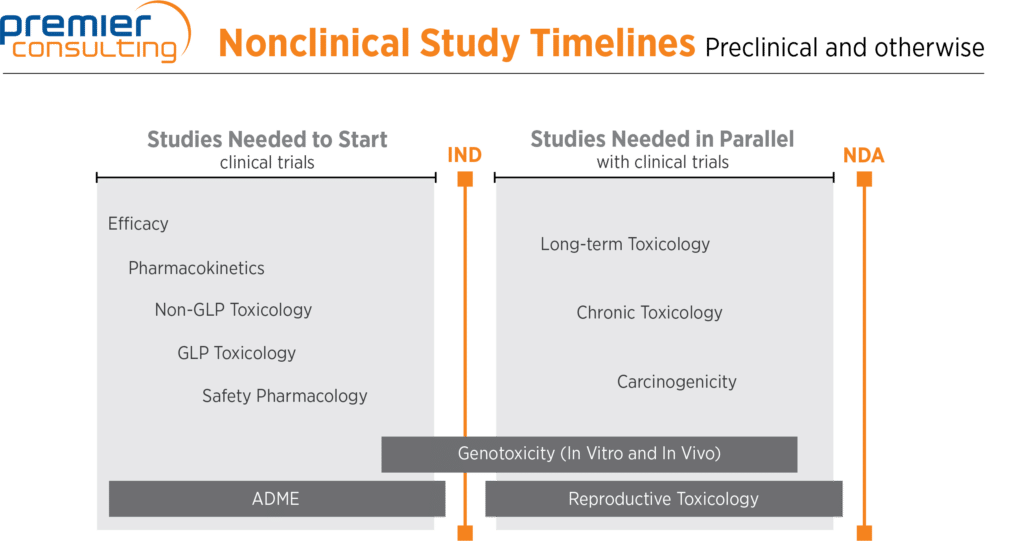
Preclinical And Nonclinical Studies—What Is The Difference, And Where In Your Program Should They Fall? | Premier Consulting

Regulatory compliance: How to shape a non-clinical development program and paediatric requirements - YouTube

Steps of non-clinical studies in drug development process. GLP: Good... | Download Scientific Diagram
Non-clinical safety studies for biotechnologically-derived pharmaceuticals: conclusions from an International Workshop - Susan A Griffiths, Cyndy E Lumley, 1998