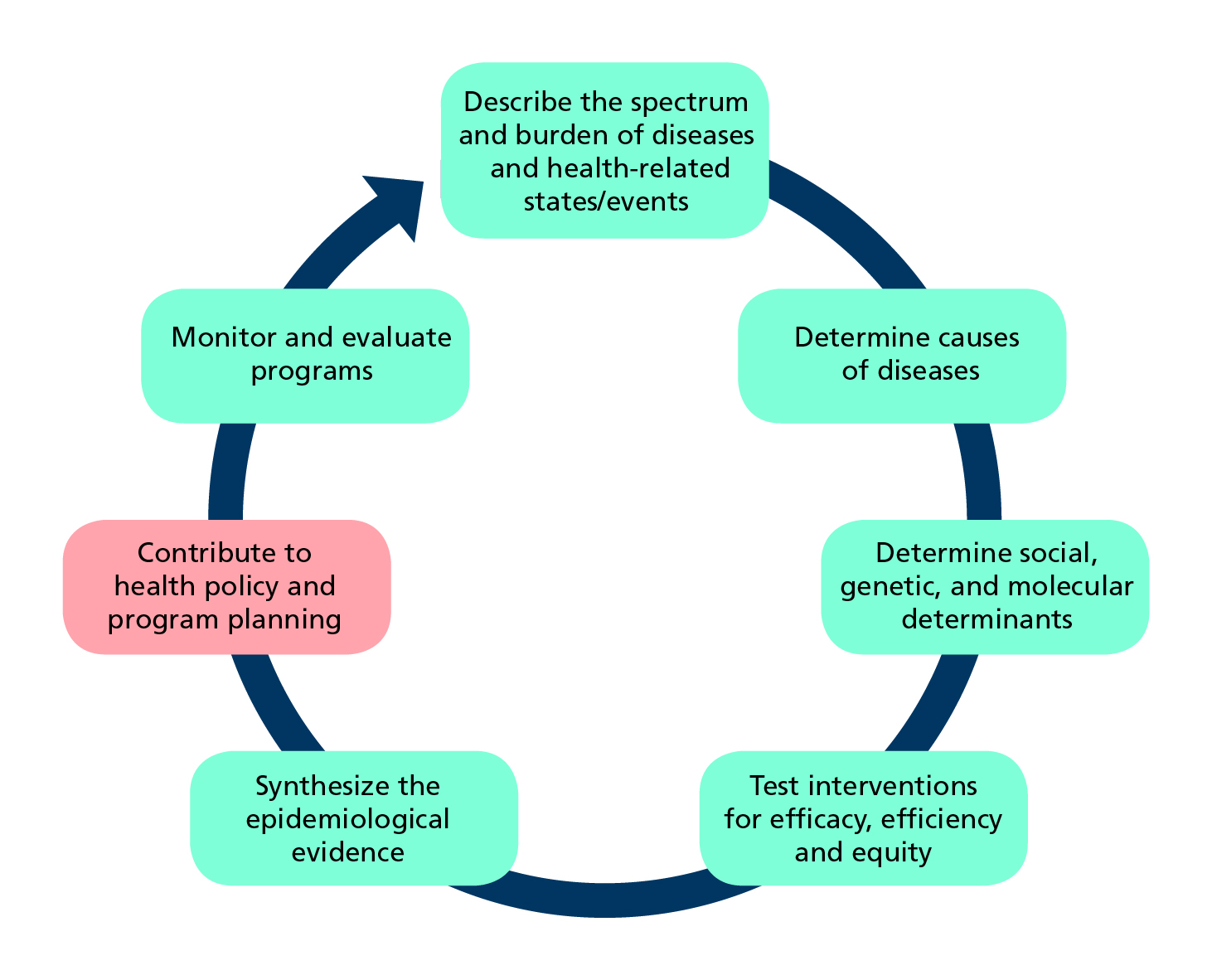
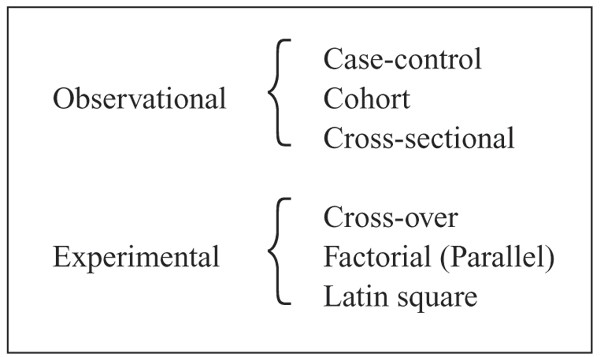
Applying Statistics to Trial Design: Sample Size, Randomization, and Control for Multiple Hypotheses | Basicmedical Key

Leveraging electronic health records for data science: common pitfalls and how to avoid them - The Lancet Digital Health

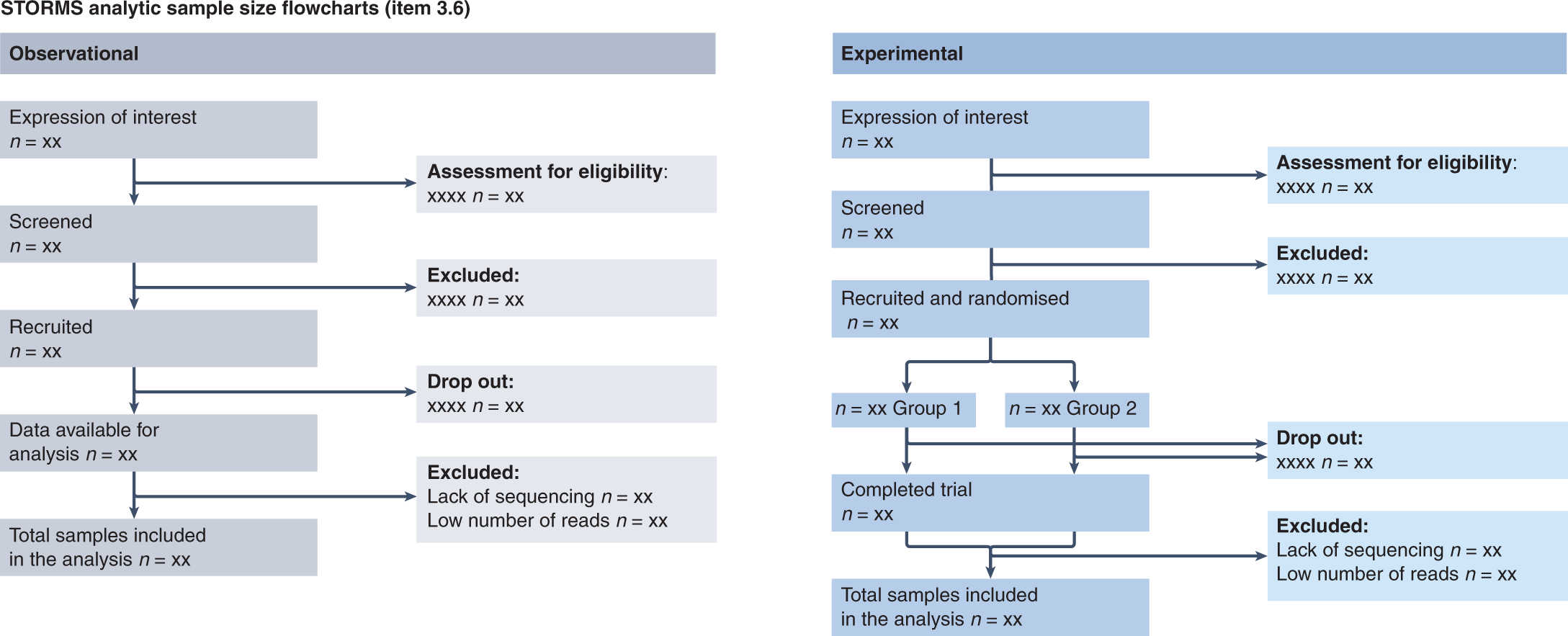
Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and Elaboration. Translation to Russian - Vandenbroucke - Digital Diagnostics

An opinion on Wastewater-Based Epidemiological Monitoring (WBEM) with Clinical Diagnostic Test (CDT) for detecting high-prevalence areas of community COVID-19 infections - ScienceDirect

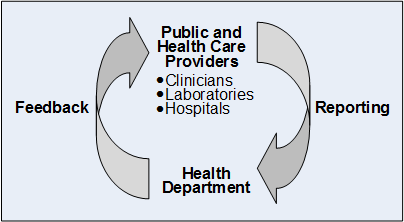
Population Point Prevalence of SARS-CoV-2 Infection Based on a Statewide Random Sample — Indiana, April 25–29, 2020 | MMWR
Epidemiological Studies Based on Small Sample Sizes – A Statistician's Point of View | Acta Veterinaria Scandinavica | Full Text

Sample size determination: A practical guide for health researchers - Althubaiti - 2023 - Journal of General and Family Medicine - Wiley Online Library

Sample Sizes for Clinical, Laboratory and Epidemiology Studies - Kindle edition by Machin, David, Campbell, Michael J., Tan, Say Beng, Tan, Sze Huey. Health, Fitness & Dieting Kindle eBooks @ Amazon.com.
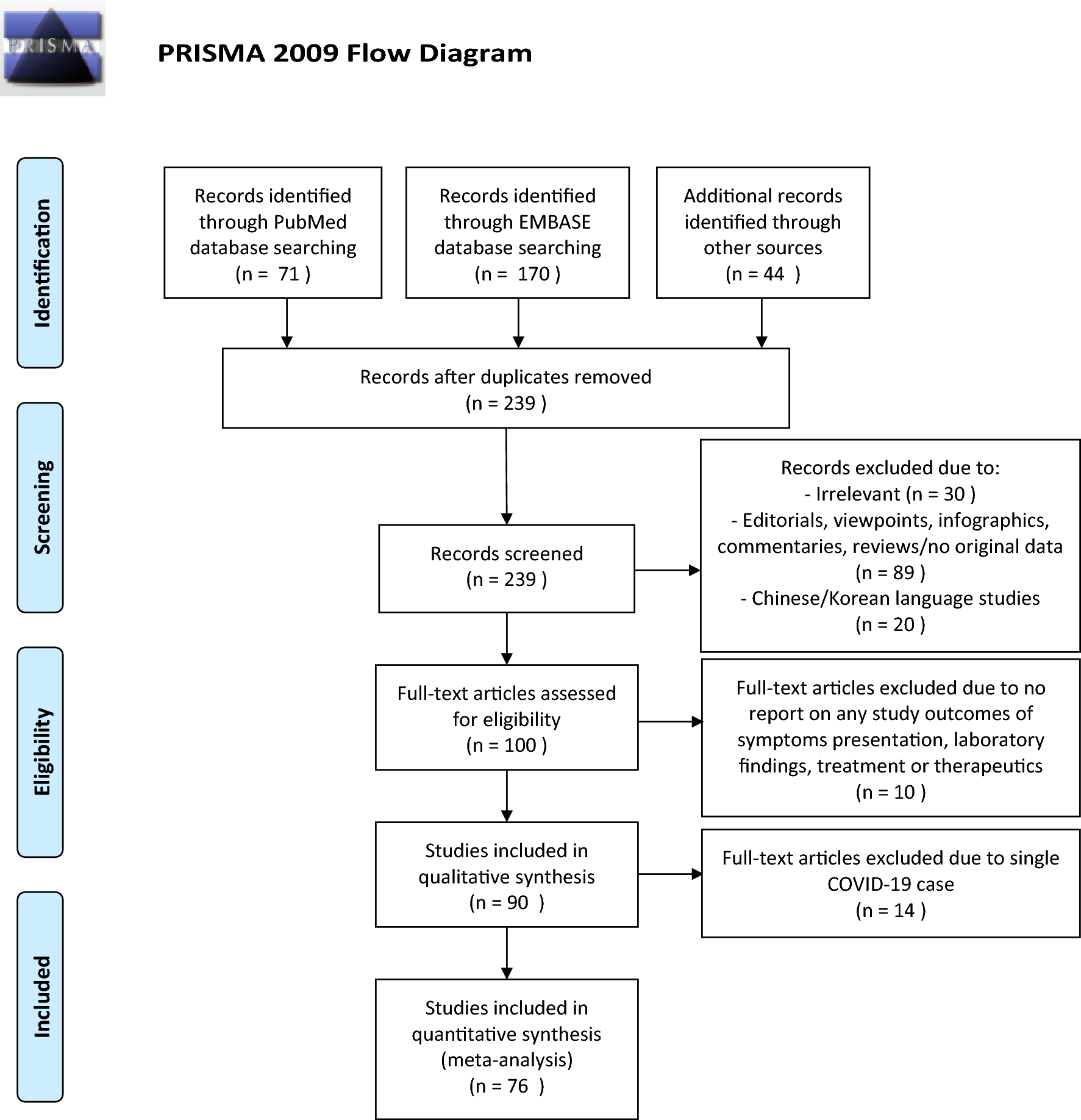
Clinical presentations, laboratory and radiological findings, and treatments for 11,028 COVID-19 patients: a systematic review and meta-analysis | Scientific Reports

Epidemiology, clinical characteristics, and virologic features of COVID-19 patients in Kazakhstan: A nation-wide retrospective cohort study - The Lancet Regional Health – Europe
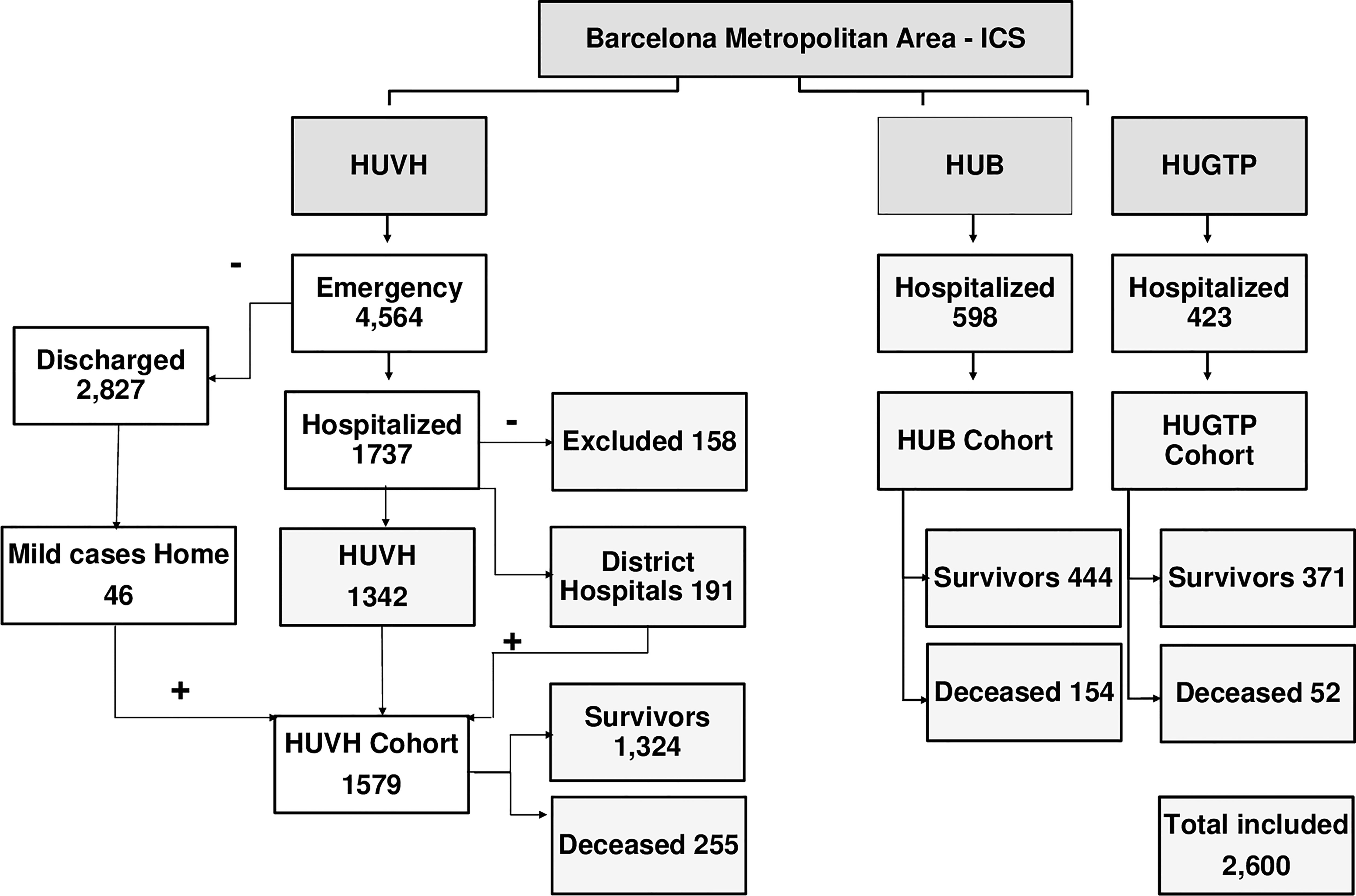
Frontiers | Exposing and Overcoming Limitations of Clinical Laboratory Tests in COVID-19 by Adding Immunological Parameters; A Retrospective Cohort Analysis and Pilot Study

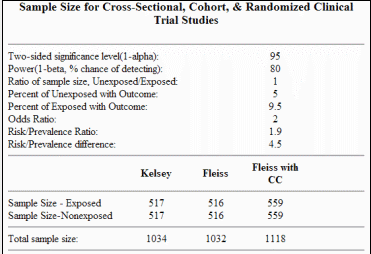
![PDF] Sample size estimation in diagnostic test studies of biomedical informatics | Semantic Scholar PDF] Sample size estimation in diagnostic test studies of biomedical informatics | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/596c4b9e0b7817120acb917cff4728fab9f95ca8/4-Table1-1.png)